A pancreatic pseudocyst is a collection of pancreatic juice encased by reactive granulation tissue (not epithelial tissue) in or around the pancreas. Pseudocysts can be single or multiple, small or large, and can be located either within or outside the pancreas. Most pseudocysts communicate with the pancreatic ductal system and contain high concentrations of digestive enzymes such as amylase and lipase.
The walls of pseudocysts are formed by adjacent structures such as the stomach, transverse mesocolon, gastrocolic omentum, and pancreas. The lining of pancreatic pseudocysts consists of fibrous and granulation tissue; the lack of an epithelial lining distinguishes pseudocysts from true pancreatic cysts.
The mechanism by which a pseudocyst is formed is necrosis and liquefaction of the pancreatic necrosis of pancreatic or peripancreatic tissue.
Pseudocyst can be seen in the following situations:
- After an episode of acute pancreatitis (in 10% of patients) - necrosis of peripancreatic tissue progresses to liquefaction and pseudocyst formation. Alternatively, a pseudocyst may result from parenchymal necrosis leading to the complete ductal disruption, and gross leakage of pancreatic juice.
- In patients with chronic pancreatitis – pseudocysts may develop after acute attacks of pancreatitis or after the pancreatic duct is obstructed. The latter causes increased intraductal pressure and leakage of pancreatic juice.
- After blunt or penetrating abdominaltrauma (including iatrogenic injuries) – The injury can directly disrupt the duct and causes leakage.
Clinical manifestations and complications of pancreatic pseudocysts include:
- Expansion of the pseudocyst can produce abdominal pain, duodenal or biliary obstruction, vascular occlusion, or fistula formation into adjacent viscera, the pleural space, or pericardium.
- Spontaneous infection.
- Digestion of an adjacent vessel can result in a pseudoaneurysm, which can produce a sudden, painful expansion of the cyst or gastrointestinal bleeding due to bleeding into the pancreatic duct.
- Pancreatic pleural effusion -can result from disruption of the pancreatic duct with fistulization into the chest.
- Pancreatic peritonitis - can be caused by disruption of the pancreatic duct with fistulisation to the abdomen.
NOTE - up to 40% of pseudocysts resolve without intervention; however, they can produce a wide range of clinical problems depending on the location and extent of the fluid collection and the presence of infection.
Pancreatitis pseudocysts are diagnoses with CT or ultrasound scan. Where the diagnosis is in doubt, the content can be aspirated (under endoscopic ultrasonography or CT scan) and examined.
The old rule mentioning that intervention is needed if the cysts are larger than 6 cm or persist beyond 6 weeks is no longer in use, and surgical intervention should be considered if any of the following is present:
- Compression of large vessels (clinical symptoms or seen on CT scan)
- Gastric or duodenal outlet obstruction
- Stenosis of the common bile duct due to compression
- Infected pancreatic pseudocysts
- Hemorrhage into pancreatic pseudocyst
- Pancreatico-pleural fistula
- Pancreatic pseudocysts and symptoms:
- Early satiety
- Nausea and vomiting
- Pain
- Upper gastrointestinal bleeding
- Asymptomatic pancreatic pseudocyst AND either of the following:
- Pseudocysts> 5cm, unchanged in size and morphology for more than 6 weeks
- Diameter> 4cm and extrapancreatic complications in patients with chronic alcoholic pancreatitis
- Suspected malignancy
Surgical drainage is the criterion standard against which all other interventions are measured in terms of success rate, mortality and recurrence rate. In recent years, however, endoscopic drainage has been introduced and can be applied provided that the cyst is near the stomach or duodenal wall:
There are two main types of endoscopic drainage:
- Transmural drainage: in this method, using endoscopy, a small incision is made in the stomach (endoscopic cystgastrostomy [ECG]) or in duodenum (endoscopic cystduodenostomy [ECD]) to let the pseudocyst drain into the stomach or duodenum. ECD is preferred over ECG.
NOTE – pseudocysts should have a mature capsule (wall thickness>3mm and <1cm), bulge the lumen and have minimum size of 5-6 cm to become eligible for endoscopic drainage.
- Transpapillary drainage: this method is safer and more effective than transmural drainage, but requires that the cyst communicates with the pancreatic duct because this method includes entering the pancreatic duct by ERCP, and from there, into the pseudocyst. Stents may be left in place to facilitate drainage.
Generally, endoscopic drainage methods are preferred over open surgical treatment if eligibility is met and there is no contraindication because these methods are less invasive and associated with fewer complications.
Laparotomy with cyst excision and internal and external drainage is still the gold standard management option; however it is ONLY considered first-line therapy for surgical intervention in the following conditions:
- Complicated pseudocysts i.e. infected or necrotic
- Pseudocysts associated with pancreatic duct stricture and a dilated pancreatic duct
- Suspected cystic neoplasia
- Presence of pseudoaneurysm, unless it has been embolised before the procedure
- Coexistence pseudocysts and bile duct stenosis
- Complications such as compression of the stomach or the duodenum, perforation or pseudoaneursyms
NOTE – pseudoaneurysm is an absolute contraindication to endoscopic drainage unless it is embolised prior to the procedure.
Generally, patients with symptomatic pseudocysts should undergo interventional measures for pseudocyst drainage. The procedure of choice is endoscopic drainage. This cyst is 10 cm in size (>5cm) and amenable to endoscopic drainage either by endoscopic transmural or transpapillary drainage. Transpapillary drainage has the lowest complication rate of all the mentioned procedures and is the method of choice if the pseudocyst communicates with the pancreatic duct. Fortunately, 80% of pseudocysts communicate with the pancreatic duct.

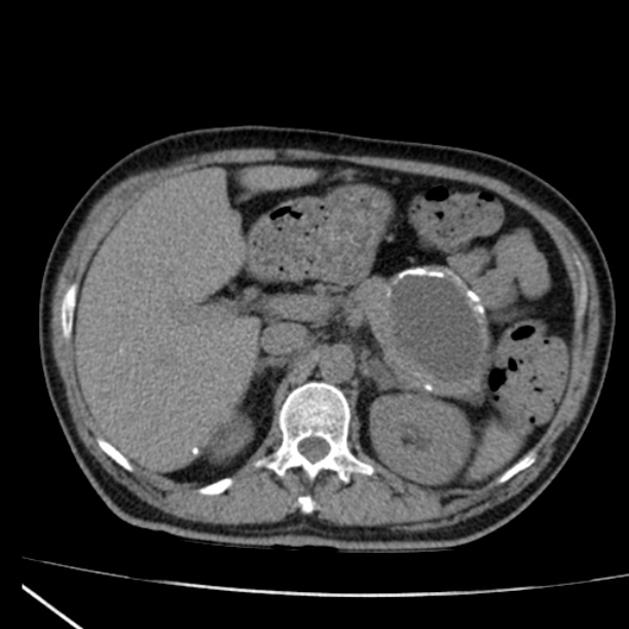
:
thanks for the point. That was great.
:
hiow often do you post in here?